 The lower the energy level of the electron, the more likely it is to absorb a photon. When an electron absorbs a photon, its energy level increases by an amount equal to the photon’s energy. Absorption spectra are also determined by the energy levels of an atom’s electrons. The emission spectrum of an atom is a plot of the intensities of all the different wavelengths of light that it emits.Īn absorption spectrum occurs when an atom absorbs light instead of emitting it. The higher the energy level of the electron, the shorter the wavelength of light that it will emit. These wavelengths correspond to the energy levels of the atom’s electrons. 
When an atom emits light, it does so at certain wavelengths. The main difference between emission and absorption spectra is that emission spectra are always continuous, while absorption spectra can be either continuous or discrete. Emission spectra occur when an atom emits light, while absorption spectra occur when an atom absorbs light. The difference between emission and absorption spectraĮmission spectra and absorption spectra are two types of atomic spectra. The strength of the absorption depends on the amount of material, the type of atoms or molecules, and the wavelength of the light. These dark lines correspond to the absorption of light at certain wavelengths by the atoms or molecules of the material through which the light passes. What is an absorption spectrum?Īn absorption spectrum is a spectrum in which there are one or more dark lines or bands. An emission spectrum is sometimes also called an atomic emission spectrum or a molecular emission spectrum. The word “emission” comes from the Latin word for “to send out”. The emission spectrum of a particular element or molecule can be used as a fingerprint to identify it. The intensity of the light at each wavelength is proportional to the number of atoms or molecules emitting at that wavelength. What is an emission spectrum?Īn emission spectrum is a type of spectrum that results from the emission of light by atoms or molecules. Emission spectra involve examining light that is emitted from molecules or atoms when energy is added, while Absorption Spectra involves looking at how light interacts with molecules or atoms when energy is removed. What is the difference between absorbance and adsorption?Įmission and absorption spectra are both unique tools used to examine the composition of atoms and elements.What is the difference between absorption and absorbance?.What is the difference between electron absorption and emission?. 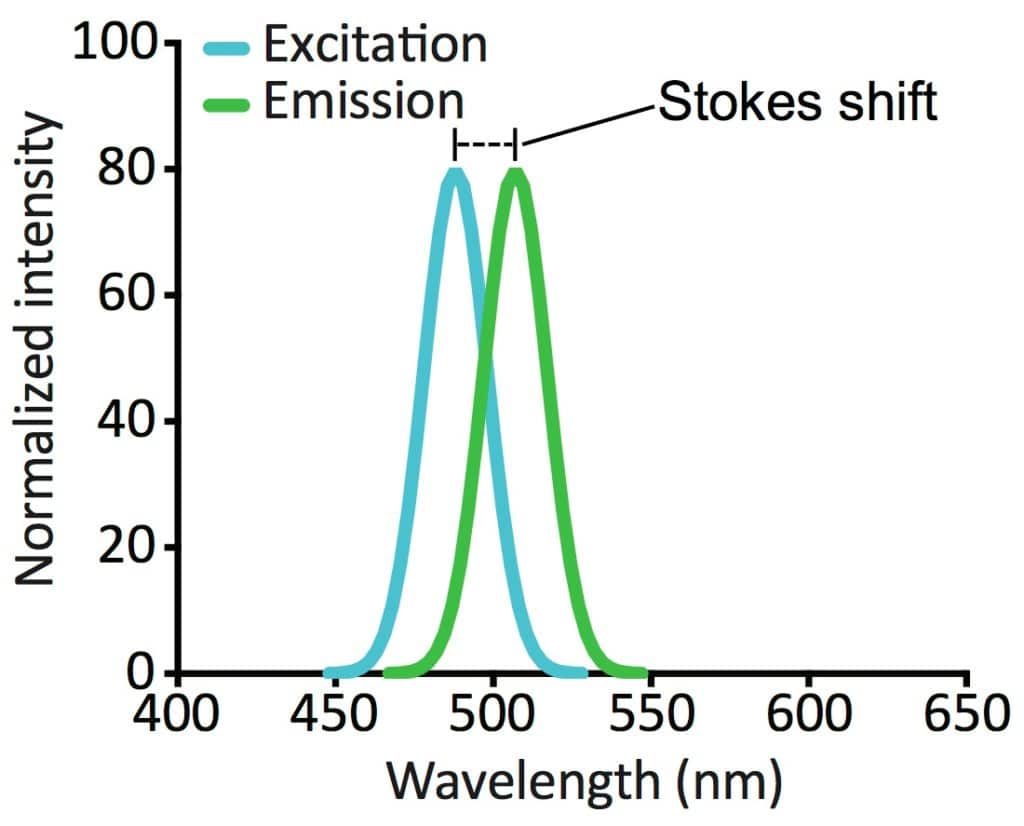
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
